Silver sits in jewelry boxes, lines the circuits of smartphones, and backs the solar panels reshaping the global energy grid. It has served as money for thousands of years and remains one of the most actively traded precious metals on earth. But most people who buy, wear, or invest in silver never stop to ask a remarkably basic question: where does silver come from?
The short answer is that virtually every atom of silver on our planet was forged inside dying stars and then concentrated into mineable deposits by volcanic activity deep within the Earth’s crust. The longer answer involves exploding stars, superheated fluids, and billions of years of geological processes that turned scattered atoms into the gleaming metal we recognize today. Understanding this journey matters for investors and collectors alike, because it explains why silver is rare enough to be precious but abundant enough to be useful.
Born in the Heart of Dying Stars
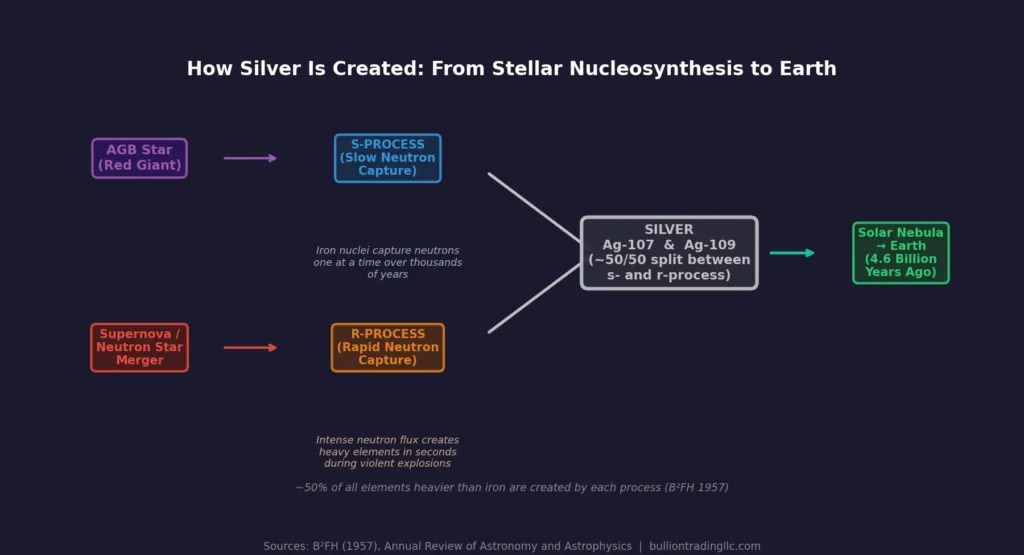
Silver’s story begins long before the Earth existed. Like all elements heavier than iron, silver cannot be created by the normal fusion reactions that power stars during their lifetimes. Instead, silver isotopes are produced through two distinct processes in stellar nucleosynthesis.
The first is the s-process (slow neutron capture), which takes place inside aging red giant stars known as asymptotic giant branch (AGB) stars. Over thousands of years, iron nuclei in these stars slowly capture free neutrons one at a time, gradually building heavier elements including silver. The process is “slow” because there is enough time between neutron captures for radioactive decay to occur, stepping the nucleus up the periodic table in an orderly fashion.
The second pathway is the r-process (rapid neutron capture), which happens during the violent explosion of a supernova or during neutron star mergers. In these events, the neutron flux is so intense that nuclei capture multiple neutrons in fractions of a second. Both of silver’s stable isotopes, Ag-107 and Ag-109, are produced through these processes, roughly split between s-process and r-process contributions.
When these stars died and expelled their material into space, the silver atoms mixed into the vast clouds of gas and dust that would, about 4.6 billion years ago, collapse to form our solar system. Every silver coin, bar, or piece of jewelry you have ever held contains atoms forged in the interior of a star that no longer exists.
From Space Dust to Earth’s Crust
Once the Earth formed and differentiated into layers, most of the planet’s heaviest elements, including silver, sank toward the core along with iron. The silver that we can actually access today is present in the Earth’s crust at a concentration of just 0.08 parts per million, roughly the same abundance as mercury. That makes silver scarce enough to be precious, but the real question is how those widely dispersed atoms became concentrated into deposits rich enough to mine.
The answer, in almost every case, involves volcanoes.
According to the geological literature, most true silver deposits, as opposed to argentiferous deposits of other metals, originated from Tertiary-era volcanism, the period of intense volcanic activity that shaped much of the Earth’s current landscape between roughly 66 million and 2.6 million years ago. This volcanic activity drove the hydrothermal processes that concentrated silver from trace amounts in ordinary rock into the rich veins and ore bodies miners have exploited for over 5,000 years.
How Volcanoes Create Silver Deposits
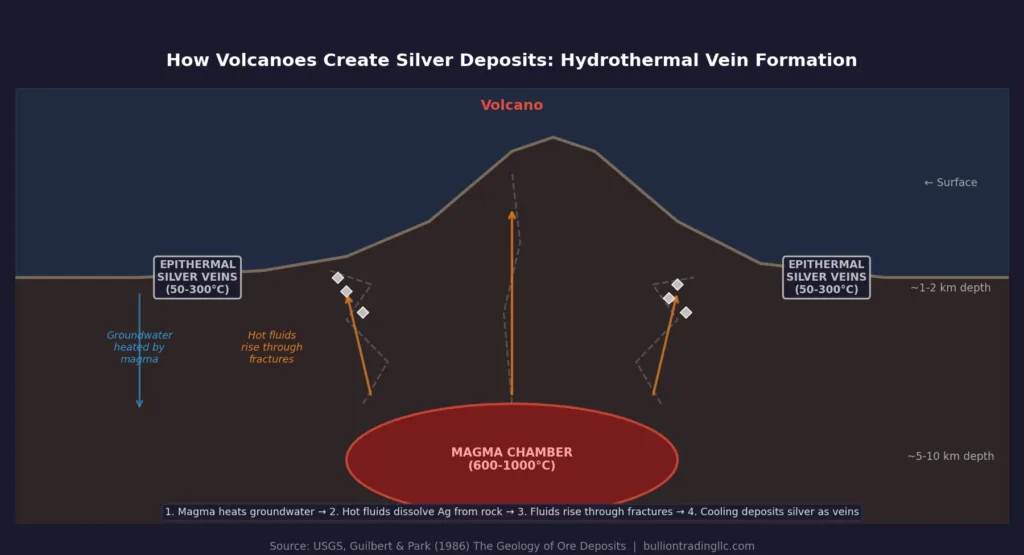
The mechanism by which volcanoes concentrate silver is a process called hydrothermal circulation, and it works like a giant, slow-motion chemistry experiment. When magma intrudes into the upper crust beneath a volcano, it heats the surrounding groundwater to extreme temperatures, sometimes exceeding 300 degrees Celsius. This superheated water becomes a powerful solvent, capable of dissolving metals, including silver, from the surrounding rocks.
These metal-laden fluids rise through cracks, faults, and fractures in the rock, driven upward by heat and pressure. As they move toward the surface and cool, the dissolved metals can no longer stay in solution. They precipitate out and deposit along the walls of the fractures, forming the mineral veins that prospectors have sought for centuries. Silver typically precipitates as sulfide minerals, especially acanthite (Ag2S) and argentite, along with native silver in some reducing environments.
The most important type of silver deposit is the epithermal vein, which forms at relatively shallow depths (within a few kilometers of the surface) and at temperatures between about 50 and 300 degrees Celsius. Geologists classify these into two subtypes. Low-sulfidation epithermal deposits form from near-neutral pH fluids that have mixed with groundwater, and they tend to produce classic bonanza-style silver veins with very high grades. High-sulfidation epithermal deposits form from more acidic, sulfur-rich fluids closer to the volcanic vent itself, and while they are often lower grade, they can be extremely large in tonnage.
This is why the world’s richest silver districts almost always sit along volcanic belts. The Ring of Fire, the horseshoe-shaped zone of intense volcanic and seismic activity encircling the Pacific Ocean, hosts a vastly disproportionate share of the planet’s silver wealth. Mexico, Peru, Bolivia, and Chile, all located along this volcanic arc, have dominated global silver production for the past 500 years and continue to do so today.
Silver as a Byproduct: The Modern Mining Reality
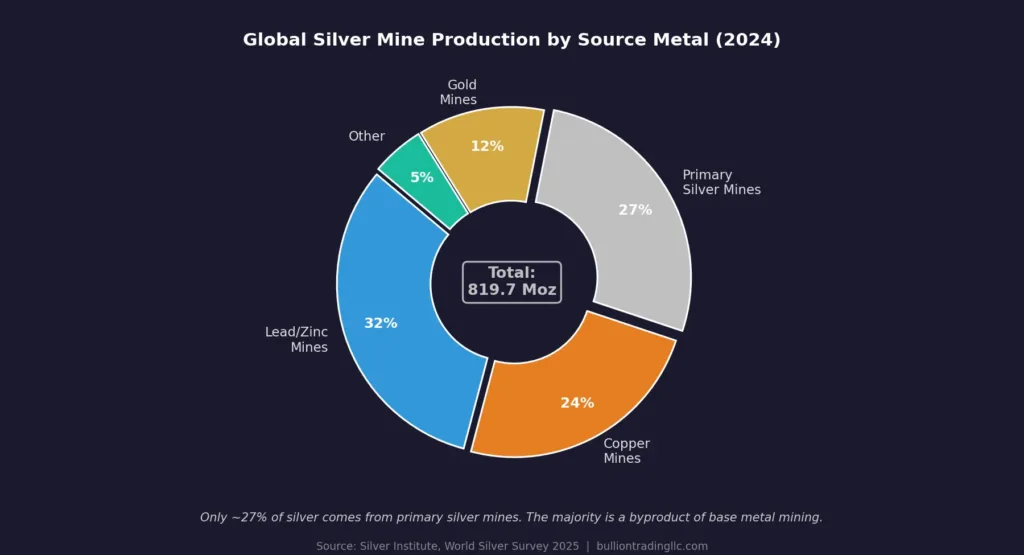
Here is something that surprises many people: the majority of the world’s silver is not actually mined from dedicated silver mines. Today, silver is primarily produced as a secondary byproduct of mining other metals, especially copper, lead, and zinc. According to the Silver Institute’s World Silver Survey 2025, silver production from lead and zinc mines remained the dominant source of global supply in 2024, though output from gold mines recorded the strongest growth at 12% year-over-year.
This happens because the same hydrothermal processes that concentrate silver also concentrate these other metals. Silver frequently occurs alongside galena (lead sulfide), chalcopyrite (copper iron sulfide), and sphalerite (zinc sulfide) in polymetallic hydrothermal veins. During the refining of these base metals, silver is recovered from the “anode slime” that collects during electrolytic refining of copper, or through the Parkes process applied to lead bullion.
In 2024, global silver mine production rose 0.9 percent to 819.7 million ounces, with Mexico remaining the leading producer, followed by China, Peru, Bolivia, and Chile. Fresnillo’s Penasquito mine in Mexico returned to full production, and additional growth came from Bolivia and the United States. An additional 193.9 million ounces came from recycling, a 12-year high, reflecting strong industrial scrap recovery and elevated silver prices encouraging sales of silverware in Western markets.
This byproduct dynamic has a significant implication for silver investors. Because most silver production depends on the economics of mining copper, lead, and zinc rather than silver itself, silver supply does not respond to price changes the way you might expect. If copper prices fall and copper mines cut production, silver supply falls too, regardless of silver prices. This structural feature makes silver supply relatively inelastic and is one reason many analysts believe the metal is chronically undervalued.
5,000 Years of Following the Volcanoes
Humans figured out where silver comes from, at least in practical terms, a very long time ago. The Silver Institute notes that silver was first mined around 3,000 BCE in Anatolia, in what is now Turkey, a region sitting directly on one of the world’s most active tectonic boundaries. By about 1,200 BCE, the center of silver production had shifted to the Laurium mines near Athens, Greece, where silver extracted from lead ores helped finance the Athenian navy and fund the democracy that shaped Western civilization.
The pattern has repeated itself throughout history. The Romans extracted silver from volcanically formed deposits in Spain, reaching peak production of 200 tonnes per year. When Spanish conquistadors arrived in the Americas after 1492, they found staggering silver deposits in Mexico, Peru, and Bolivia, all products of the same hydrothermal processes at work along the Ring of Fire. Between 1500 and 1800, those three countries accounted for over 85% of global silver production, with the legendary mines at Potosi and Zacatecas both epithermal deposits formed by ancient volcanic activity.
Even today, the connection between volcanoes and silver production remains as strong as ever. The world’s top producing mines, including Cannington in Australia, Fresnillo in Mexico, and San Cristobal in Bolivia, all operate in geological settings created by ancient or ongoing volcanism.
Why This Matters for Silver Investors
Understanding where silver comes from is more than geological trivia. It provides important context for assessing silver’s long-term value and supply outlook.
Silver cannot be manufactured or synthesized economically. Every ounce that enters the market must be mined from deposits that took millions of years to form or recycled from existing products. The geological processes that create silver deposits are extraordinarily slow, and the richest, most accessible deposits have been mined for centuries. New discoveries are increasingly found at greater depths and in more remote locations, translating to higher production costs.
Meanwhile, industrial demand for silver continues to set records. Total global demand reached 1.16 billion ounces in 2024, driven primarily by the photovoltaic sector, electronics, and automotive applications, with AI adding a new layer of demand.
When a metal’s supply is geologically constrained and largely produced as a byproduct, while its demand is accelerating due to industrial megatrends, the long-term investment case becomes difficult to ignore.

