If you own silver bullion, you have probably noticed it: that dull, dark film that slowly creeps across your once-brilliant coins or bars. Silver tarnish is one of the most common concerns among precious metals investors, and for good reason. Nobody wants to pull a prized American Silver Eagle out of storage only to find it looking like it spent a decade buried in a garden. But here is the good news. Tarnish is a well-understood chemical process, it does not reduce the metal’s intrinsic value, and it is largely preventable with the right storage practices.
Whether you are holding silver coins and bars as a long-term hedge or actively trading, understanding why silver tarnishes and how to deal with it protects both the appearance and the resale premium of your investment.
The Chemistry Behind Silver Tarnish
Silver tarnish is not rust, and it is not corrosion in the way most people think of it. What actually happens is a chemical reaction between silver and sulfur-containing compounds in the environment. When silver (Ag) comes into contact with hydrogen sulfide (H2S), a gas present in trace amounts in the air around us, it forms silver sulfide (Ag2S), the dark layer you see on tarnished silver.
The reaction itself is straightforward:
2 Ag + H2S → Ag2S + H2
Unlike iron oxide (rust), which is structurally destructive, silver sulfide forms a thin surface layer that does not penetrate or degrade the silver underneath. Silver is classified as a noble metal precisely because it does not react with oxygen in air, even at high temperatures. It is specifically the sulfur compounds, not oxygen, that cause tarnish.
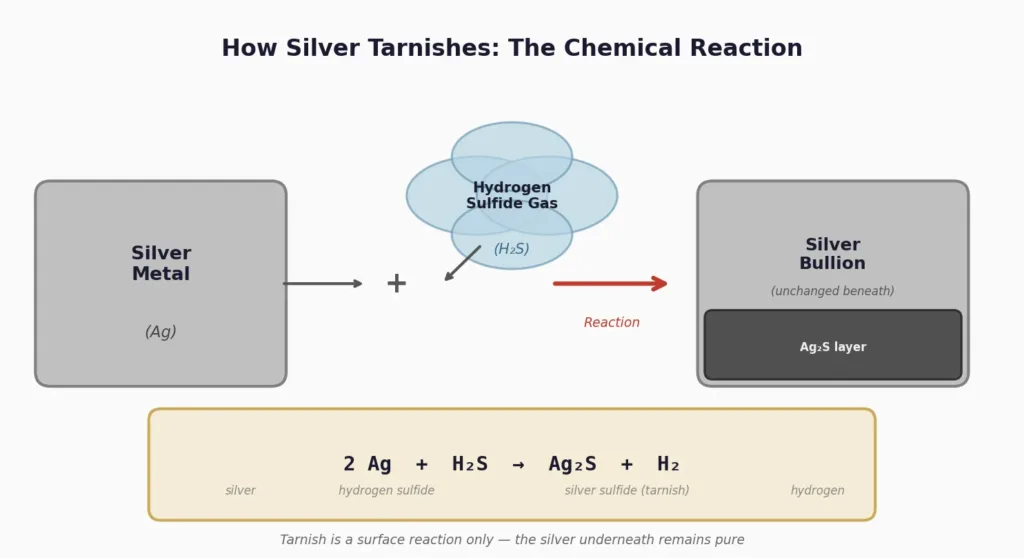
This is an important distinction for bullion investors. Tarnish is purely cosmetic. A tarnished silver bar contains exactly the same amount of fine silver as the day it was minted. Refiners and dealers who buy back bullion price on metal content, not shininess, so tarnish alone does not reduce melt value.
What Makes Silver Tarnish Faster?
Not all storage environments are created equal. Several factors dramatically accelerate the tarnishing process, and understanding them is the first step toward keeping your bullion looking its best.
Humidity is the single biggest accelerator. Moisture in the air acts as a catalyst for the silver-sulfur reaction. Research from the National Institute of Standards and Technology (NIST) has shown that elevated humidity significantly increases the rate of silver sulfide formation. If you live in a coastal or tropical climate, your silver will tarnish noticeably faster than in a dry region.
Airborne pollutants also play a major role. Hydrogen sulfide exists naturally in the atmosphere at low concentrations, but certain environments push those levels much higher. Proximity to industrial areas, volcanic regions, or even heavy traffic increases sulfur compound concentrations. Household sources matter too: rubber bands, certain paints, wool, felt, and natural gas from stoves all release sulfur gases that accelerate tarnish.
Direct skin contact is another trigger many collectors overlook. Human sweat contains salts, oils, and trace sulfur compounds that react with silver on contact. Handling coins with bare hands leaves behind fingerprint residues that tarnish faster than the surrounding surface, creating uneven discoloration. This is especially damaging on proof coins where surface quality directly affects value.
Certain storage materials can be surprisingly problematic. Cardboard, newspaper, rubber bands, and PVC-based plastic flips all emit sulfur or chlorine gases that promote tarnishing. Even some wood types, particularly oak, release acetic acid vapors that damage silver over time.
Does Tarnish Affect Silver Bullion Value?
This is the question that keeps new investors up at night, and the answer depends on what type of silver you are holding. For standard bullion products like generic rounds, bars, and common-date government coins, tarnish has essentially zero effect on the melt value. A 10-ounce silver bar with heavy tarnish contains the same amount of silver as a freshly minted one, and any reputable dealer prices buybacks on metal content.
However, for numismatic or collectible coins, the situation is different. Proof coins, limited mintage pieces, and coins graded by PCGS or NGC can lose significant premium if tarnished or improperly cleaned. A coin graded MS-70 that develops tarnish may be regraded lower, costing substantial premium over spot. In these cases, prevention is worth far more than cure.
It is worth noting that some collectors actually prize a certain kind of natural tarnish called “toning”. Attractive rainbow or deep blue toning that develops evenly over decades can increase a coin’s desirability at auction, particularly on American Silver Eagles and Morgan Silver Dollars. The distinction is largely subjective, but generally, even colorful toning is valued while blotchy dark spots are not.
How to Prevent Silver Tarnish on Bullion
Prevention is always better than remediation, and when it comes to silver bullion, a few simple practices can keep your holdings looking pristine for decades.
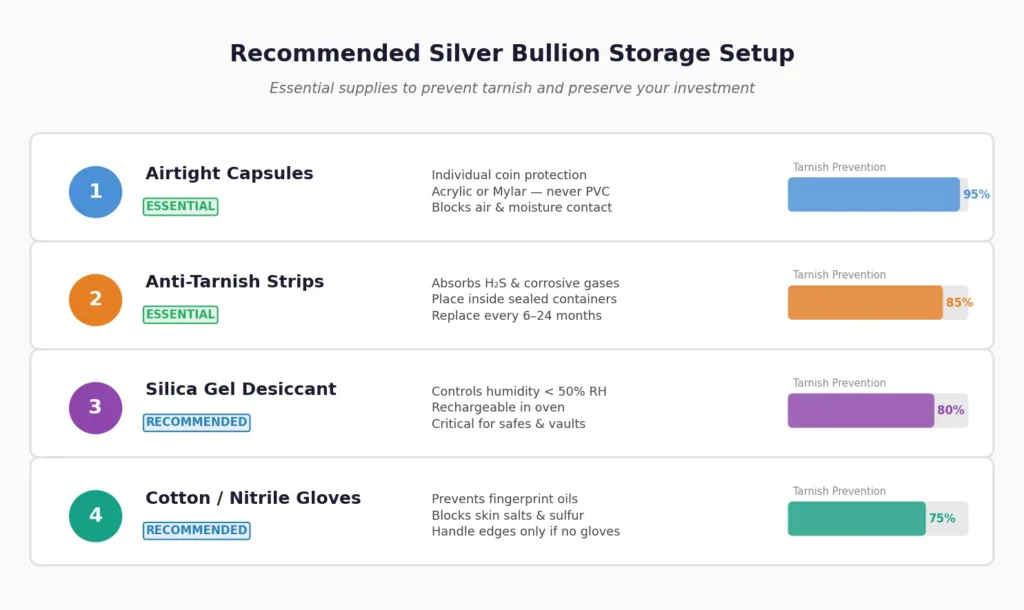
Control humidity. Store your silver in a dry environment with relative humidity below 50%. If you are using a home safe, place a rechargeable silica gel desiccant inside. These inexpensive packets absorb moisture and can be recharged by heating in an oven. For larger collections, a climate-controlled room or bank safe deposit box is ideal.
Use anti-tarnish strips and intercept technology. Anti-tarnish strips (also called vapor phase inhibitors) are chemically treated paper strips that absorb hydrogen sulfide and other corrosive gases before they reach your silver. Placing one strip inside each sealed container or tube of coins provides protection for six months to two years, depending on conditions.
Store in appropriate containers. Airtight capsules for individual coins and sealed tubes for multiples are the gold standard. Look for capsules made from inert materials like acrylic or Mylar. Avoid PVC flips at all costs, as PVC breaks down over time and releases hydrochloric acid that creates a green, sticky residue on coins known as “PVC damage”. Polyethylene or polypropylene flips are safe alternatives.
Handle with gloves. Always wear clean cotton or nitrile gloves when handling silver bullion. If gloves are not available, handle coins only by the edges, never touching the flat surfaces. This simple habit prevents fingerprint-shaped tarnish patterns that are nearly impossible to remove without damage.
Separate silver from other metals. Storing silver in contact with copper, brass, or iron can cause galvanic reactions that accelerate tarnish. Keep silver items separated, ideally each in its own capsule or sleeve.
How to Safely Remove Tarnish from Silver Bullion
If tarnish has already developed, you have several options, but caution is key. Aggressive cleaning damages surfaces, reduces numismatic value, and can leave scratches visible under magnification. The mantra in the coin collecting community is simple: when in doubt, do not clean it.
For standard bullion (non-numismatic bars and rounds), the safest method is the aluminum foil and baking soda bath. This is not a gimmick; it is real electrochemistry. Line a glass or plastic container with aluminum foil, place the tarnished silver on the foil, and pour in hot (not boiling) water with one tablespoon of baking soda per cup. The aluminum acts as a sacrificial anode, donating electrons that convert silver sulfide back into metallic silver. As the Royal Society of Chemistry explains, this method reverses the tarnish rather than removing silver from the surface.
The chemical reaction at work:
3 Ag2S + 2 Al → 6 Ag + Al2S3
This approach is preferred over polishing because it does not remove any silver metal. Polishing physically abrades the surface, removing a thin layer of silver along with the tarnish. On bullion coins, this removes original mint luster and creates visible hairline scratches that reduce a coin’s grade and premium.
For numismatic or graded coins, the answer is usually: leave them alone. Professional coin dealers and grading services widely advise against cleaning valuable coins. PCGS’s guide on coin preservation explicitly recommends against all forms of cleaning for graded coins. If a valuable coin develops tarnish, consult a professional numismatist or conservation specialist before attempting any treatment.
Avoid commercial silver dips for bullion coins. Products like silver dip solutions work by dissolving the silver sulfide layer, but they can leave a dull, matte finish and strip away the original surface character. For silverware or jewelry these products may be acceptable, but for investment-grade bullion, they are best avoided.
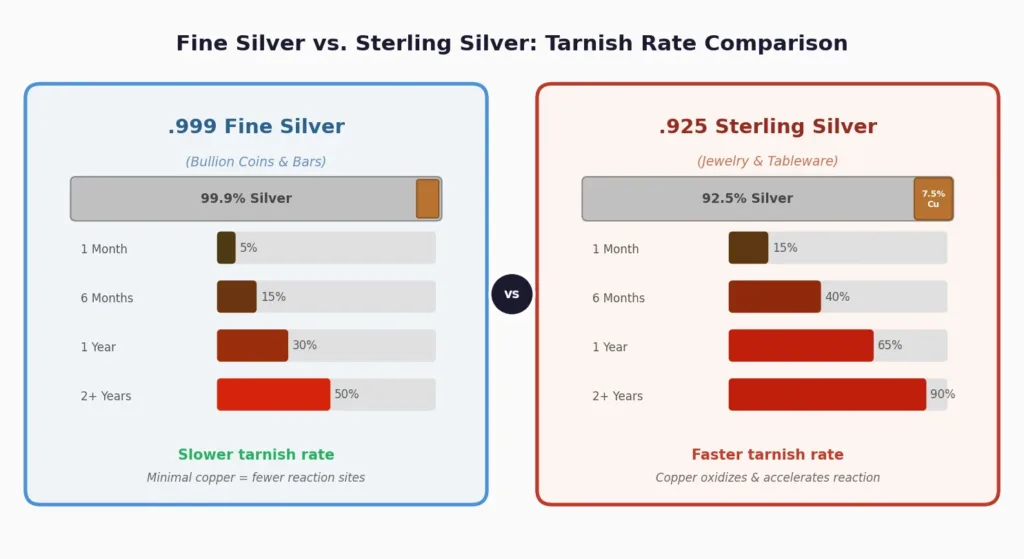
Sterling Silver vs. Fine Silver: Tarnish Differences
Not all silver tarnishes at the same rate. Most silver bullion is struck from .999 fine silver (99.9% pure), which actually tarnishes more slowly than sterling silver (92.5% silver, 7.5% copper). The copper content in sterling silver oxidizes more readily and accelerates the overall tarnishing process, which is why sterling jewelry tends to darken faster than a pure silver bullion coin.
Some modern mints have introduced special anti-tarnish finishes. The Royal Canadian Mint, for example, uses a proprietary micro-engraving and surface treatment on its Silver Maple Leaf coins that reduces tarnishing. The Perth Mint and several private mints also offer capsule-sealed coins that arrive in airtight packaging designed to preserve their appearance indefinitely.
Long-Term Storage Best Practices for Silver Investors
For investors building positions over years or decades, the long game of silver storage comes down to environment control and good habits. If you are storing at home, a quality fireproof safe in a climate-controlled room is the baseline. Add silica gel desiccants, include anti-tarnish strips, and ensure everything is sealed in appropriate capsules or tubes. Keep the safe away from kitchens, bathrooms, and laundry rooms where humidity and chemical vapors are highest.
For larger holdings, professional vault storage through dealers or allocated storage services ensures consistent climate control and security, essentially eliminating tarnish risk entirely.
One often overlooked tip: do not check on your silver too frequently. Every time you open a sealed container, you introduce fresh air, moisture, and contaminants. If you are investing for the long term, seal it properly and leave it alone.
Conclusion
Silver tarnish is a natural chemical process, not a defect or sign of impurity. It happens because silver reacts with sulfur compounds in the environment, forming a thin layer of silver sulfide on the surface. While cosmetically unpleasant, tarnish does not reduce your bullion’s precious metal content or its melt value.
The best approach is prevention: low-humidity storage, anti-tarnish strips, airtight capsules, and gloves for handling. If tarnish does develop on standard bullion, the aluminum foil and baking soda electrochemical bath is the safest removal method. For numismatic coins, resist the urge to clean and consult a professional instead.

